The electron groups have an octahedral arrangement. The Lewis structure has six electron groups around the iodine atom: four bonding pairs and two lone pairs. These steps can again be used to determine the electron-pair geometry and molecular structure of the iodine tetrachloride anion. The lone pair occupies one of the equatorial positions, and the molecule is seesaw-shaped. The electron groups have a trigonal bipyramidal geometry. 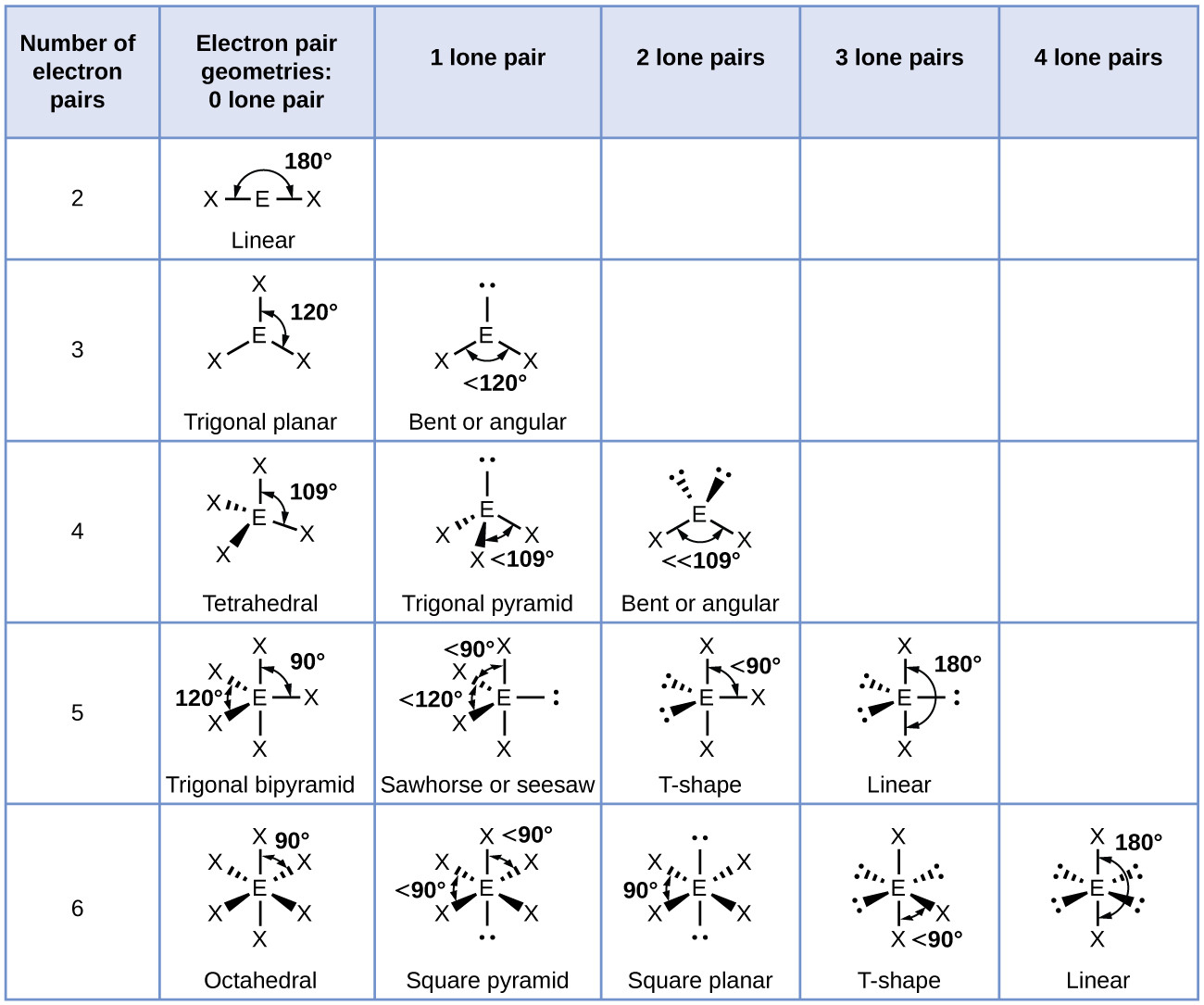
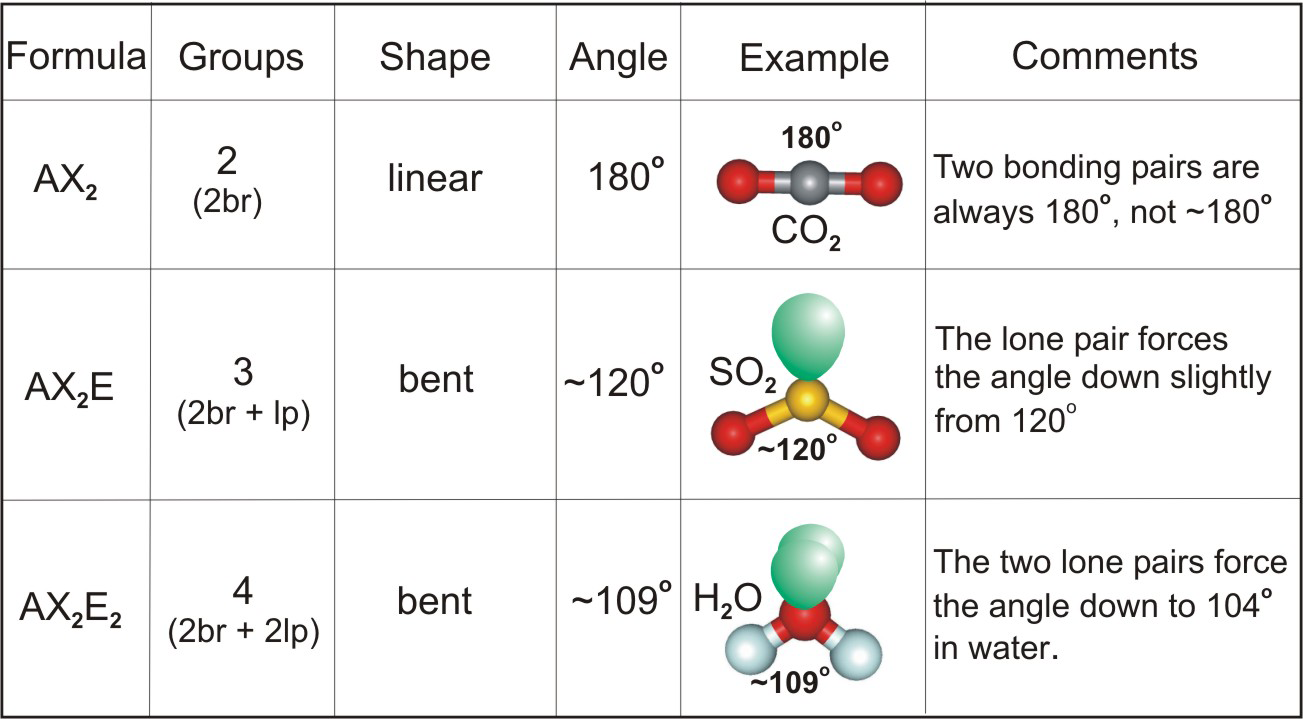
The Lewis structure of tellurium tetrachloride has five electron groups around the tellurium atom: four bonding pairs and one lone pair. The electron-pair and molecular geometries are identical because there are no lone pairs on the central atom, and carbon dioxide molecules are linear. The two-electron groups orient themselves on opposite sides of the central carbon atom with a bond angle of 180°. The Lewis structure of carbon dioxide shows the two-electron groups around the carbon atom-as each double bond counts as one electron group. The same protocol is used to predict the electron-pair geometry and molecular structure for carbon dioxide. The lone pair reduces the bond angle to less than 109.5°. However, because of the lone pair, the molecular geometry is trigonal pyramidal. The electron pair geometry is tetrahedral. Now determine the electron-pair geometry. Around phosphorus, there are four electron groups: three bonding pairs and one lone pair. Next, count the total number of electron groups on the central atom. The first step is to draw the Lewis structure of the molecule. VSEPR theory helps to determine electron-pair geometries and molecular geometries.Ī series of steps is used to predict the geometry and bond angles of molecules, such as phosphorus trichloride. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
